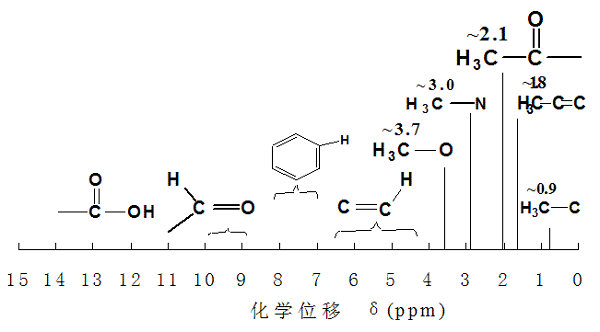
First, the development of a brief historyThe first stage: 1945 to 1951, the invention of nuclear magnetic resonance and lay the theoretical and experimental basis of the period: Bloch (Stanford University, observed in the water proton signal) and Purcell (Harvard University, observed in the paraffin proton signal) obtained Nobel bonus.The second stage: 1951 to 1960 for the development period, its role by chemists and biologists recognized, to solve many important problems. 1953 appeared in the first 30MHz nuclear magnetic resonance spectrometer; 1958 and early in the emergence of 60MHz, 100MHz instrument. In the mid-1950s, 1H-NMR, 19F-NMR and 31P-NMR were developed.The third stage: 60 to 70 years, NMR technology leap period. Pulse Fourier transform technology to improve the sensitivity and resolution, can be routinely measured 13C nuclear; dual frequency and multi frequency resonance technology;The fourth stage: the late 1970s theory and technology development mature.1,200, 300, 500 MHz and 600 MHz superconducting NMR spectrometers;2, the application of a variety of pulse series, in the application made important development;3, 2D-NMR appeared;4, multi-core research, can be applied to all magnetic cores;5, there have been “nuclear magnetic resonance imaging technology” and other new branch disciplines.Second, the main purpose:1. Determination and confirmation of the structure, and sometimes can determine the configuration, conformation2. Compound purity inspection, the sensitivity of thinner, paper chromatography high3. Mixture analysis, such as the main signal does not overlap, without separation can determine the proportion of the mixture.4. Proton exchange, the rotation of a single bond, the transformation of the ring and other chemical changes in the speed of the presumption1. the spin of the nucleusOf the isotopes of all elements, about half of the nuclei have spin motion. These spin nuclei are the object of nuclear magnetic resonance. Spin Quantum: The number of quantum numbers describing the spin motion of the nucleus, which can be an integer, a half integer, or a zero.In the organic compound composition elements, C, H, O, N is the most important element. In its isotopes, 12C, 16O are non-magnetic and therefore do not undergo nuclear magnetic resonance. 1H natural abundance of large, strong magnetic, easy to determine, so the NMR study was mainly for the proton. 13C abundance is small, only 12C 1.1%, and the signal sensitivity is only a proton to get 1/64. So the total sensitivity of only 1/6000 of 1H, more difficult to determine. But in the past 30 years, nuclear magnetic resonance instrument is greatly improved, can be measured in a short time 13C spectrum, and give more information, has become the main means of NMR. 1H, 19F, 31P natural abundance of large, strong magnetic, and nuclear charge distribution of spherical, the most easy to determine.2. Nuclear magnetic resonance phenomena① Precession: Spin with a certain magnetic moment Under the action of external magnetic field H0, this core will form angle for the kinematic motion: is the precession kinematic velocity, which is proportional to H0 (external magnetic field strength).② spin nuclear in the external magnetic field orientation: no external magnetic field, the spin magnetic orientation is chaotic. The magnetic core is in the external magnetic field H0, with (2I + 1) orientation. The spin of the magnetic core in the external magnetic field can be analogous to the precession (pronation, swing) of the gyroscope in the gravitational field.③ conditions of nuclear magnetic resonanceThe magnetic resonance magnetic field must have the magnetic nuclei, the external magnetic field and the RF magnetic field. The frequency of the RF magnetic field is equal to the precession frequency of the spin nucleus, and the resonance occurs from the low energy state to the high energy state.④ nuclear magnetic resonance phenomenon:In the vertical direction of the external magnetic field H0, a rotating magnetic field H1 is applied to the precession nucleus. If the rotational frequency of H1 is equal to the rotational precession frequency of the nucleus, the precession nucleus can absorb energy from H1 and transition from low energy state to high energy state Nuclear magnetic resonance.3. Saturation and relaxationLow energy nuclear is only 0.001% higher than high energy nuclear. Therefore, the low energy state core is always more than the high energy nuclear, because such a little surplus, so can observe the absorption of electromagnetic waves. If the nuclear continuous absorption of electromagnetic waves, the original low energy state is gradually reduced, the intensity of the absorption signal will be weakened, and ultimately completely disappeared, this phenomenon is called saturation. When saturation occurs, the number of cores in the two spin states is exactly the same. In the external magnetic field, the low-energy nuclei are generally more nuclear than the high-energy state, absorb the electromagnetic wave energy and migrate to the high-energy state of the core will be released by a variety of mechanisms of energy, and return to the original low energy state, this process called relaxation.4. Shield effect – chemical shift① ideal state of resonanceFor isolated, bare nuclei, ΔE = (h / 2π) γ · H;Under certain H0, a nucleus has only one ΔEΔE = E outside = hνOnly the only frequency ν of absorptionSuch as H0 = 2.3500T, 1H absorption frequency of 100 MHz, 13C absorption frequency of 25.2 MHz② real core: shielding phenomenonNuclear outside the electron (not isolated, not exposed)In the compounds: the interatomic binding (role) is different, such as chemical bonds, hydrogen bonds, electrostatic interactions, intermolecular forcesImagine: In H0 = 2.3500 T, due to the outer electrons of the shield, in the nuclear position, the real magnetic field is slightly smaller than 2.3500 TResonance frequency slightly higher than 100 MHzHow much is it? 1H is 0 to 10, and 13C is 0 to 250The hydrogen nuclei have electrons outside, and they repel the magnetic field lines of the magnetic field. For the nucleus, the surrounding electrons are shielded (Shielding) effect. The greater the density of the electron cloud around the core, the greater the shielding effect, the corresponding increase in magnetic field strength to make it resonant. The electron cloud density around the nucleus is affected by the connected groups, so the nuclei of different chemical environments, they suffer from different shielding effects, their nuclear magnetic resonance signals also appear in different places.③ If the instrument is measured with a 60MHz or 100MHz instrument, the electromagnetic wave frequency of the organic compound proton is about 1000Hz or 1700Hz. In determining the structure, the need to determine the correct resonant frequency, often requires several Hz accuracy, generally with the appropriate compound as the standard to determine the relative frequency. The difference between the resonant frequency of the standard compound and the resonant frequency of a proton is called the chemical shift.5. H NMR spectroscopy informationThe number of signals: how many different types of protons are present in the moleculeThe position of the signal: the electronic environment of each proton, the chemical shiftThe intensity of the signal: the number or number of each protonSplit situation: how many different protons are presentThe chemical shift of common types of organic compounds① induced effect② conjugate effectThe conjugation effect is weak or enhanced by proton shielding due to the displacement of the π electrons③ anisotropic effectIt is difficult to explain the chemical shift of H with respect to pi-electrons, and it is difficult to explain the electronegativity④ H key effectROH, RNH2 in 0.5-5, ArOH in 4-7, the range of change, the impact of many factors; hydrogen bonding with temperature, solvent, concentration changes significantly, you can understand the structure and changes related to hydrogen bonds.⑤ solvent effectBenzene forms a complex with DMF. The electron cloud of the benzene ring attracts the positive side of the DMF, rejecting the negative side. α methyl is in the shielding region, the resonance moves to the high field; and β methyl is in the masking region, the resonance absorption moves to the low field, and the result is that the two absorption peak positions are interchanged.
Quelle: Meeyou Carbide